Write the formula for the binary compound iron (III) oxide.
The general rules for writing formula of binary compound are as follows:. The formula for the ionic compound formed from aluminum and chlorine should be determined. Concept Introduction: The compounds which contain ionic bond, which is a type of bond which formed between positive metal ion and negative, non-metal ions. (d). Interpretation Introduction. Interpretation: The formula for the.
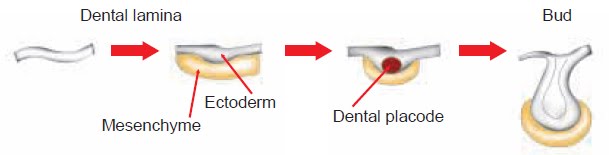
A binary compound is one made of two different elements. There can be one of each element such as in CuCl or FeO. There can also be several of each element such as Fe 2 O 3 or SnBr 4. This lesson shows you how to write the formula of a binary compound from the word name when a cation of variable charge is involved. The four formulas above are all examples of this type.
Writing Formulas for Binary Ionic Compounds. If you know the name of a binary ionic compound, you can write its chemical formula. Start by writing the metal ion with its charge, followed by the nonmetal ion with its charge. Because the overall compound must be electrically neutral, decide how many of each ion is needed in order for the positive and negative charge to cancel each other out.
The chemical formula of a compound represents this combination. The single unit can be molecules or groupings of ions. Every molecule, ion and ionic compound has a name. A system of naming chemical substances is called a chemical nomenclature. There are various ways of naming substances. The systematic naming is the most useful. A systematic system follows a set of rules. The set of rules will.
Binary Compounds of Metals with Fixed Charges Given Name, Write the Formula Return to Nomenclature menu. A binary compound is one made of two different elements. There can be one of each element such as in sodium bromide or potassium iodide. There can also be several of each element such as lithium oxide or aluminum bromide. Please remember that all elements involved in this lesson have ONLY.
Learning to Name and Write Formulas:. - Applying the rules for naming or formula writing for that type of compound. - Practicing until it becomes second nature. How to Write Chemical Formulas for Molecular Compounds. Formula writing for molecular compounds is probably the easiest type of formula writing. The key is memorizing the prefixes. Many of the prefixes you'll be familiar with since.
To name a binary molecular compound, use the following guidelines. 1. Write the names of the elements in the order listed in the formula. 2. Use prefixes appropriately to indicate the number of each kind of atom. If just one atom of the first element is in the formula, omit the prefix mono- for that element. Also, the vowel at the end of a.